Anaerobic cellular respiration and fermentation in microorganisms
I can describe the reactants and products of anaerobic cellular respiration in microorganisms, and ways in which the process is useful to us.

Lesson details
Key learning points.
- In microorganisms, anaerobic cellular respiration produces carbon dioxide and a type of alcohol called ethanol.
- A word summary of the reactants and products of anaerobic cellular respiration in microorganisms.
- Anaerobic cellular respiration in microorganisms is also called fermentation.
- Humans use fermentation in microorganisms to make useful products such as bread, yogurt and alcoholic drinks.
Common misconception
Students can often confuse the word summaries for the different types of cellular respiration.
This lesson directly compares the different word summaries for anaerobic cellular respiration to help understand the differences between them.
Microorganism - A microorganism is a living organism made up of one or a small number of cells.
Anaerobic cellular respiration - Anaerobic cellular respiration is a form of cellular respiration which occurs without oxygen.
Ethanol - Ethanol is a product of anaerobic cellular respiration in microorganisms.
Alcohol - Alcohols are a group of compounds containing carbon, hydrogen and oxygen.
Fermentation - The process of anaerobic respiration in microorganisms is called fermentation.
Content guidance
- Depiction or discussion of mental health issues
Supervision
Adult supervision required
This content is © Oak National Academy Limited ( 2024 ), licensed on Open Government Licence version 3.0 except where otherwise stated. See Oak's terms & conditions (Collection 2).
Starter quiz
6 questions.

23. Investigation into factors affecting respiration in yeast
- 00:54 Why is sucrose solution added to the yeast suspension?
- 01:12 Why must the syringe have a weight on top?
- 01:25 What gas is given off?
- 01:30 How could you improve the precision of this experiment?
yeast suspension (100g/dm 3 )
Sucrose solution (0.4moldm -3 )
thermometer
access to hot and cold water
1dm 3 beaker for carrying water
20cm 3 syringe
| Hazard | Risk | Control measure |
|---|---|---|
Yeast solution - irritant to eyes | Contact with eyes | Eye: flood with tap water (10min) |
SITEMAP * HOME PAGE * SEARCH * UK KS3 level Science Quizzes for students aged ~13-14
UK GCSE level Biology * Chemistry * Physics ~14-16 * Advanced pre-university Chemistry ~16-18
UK GCSE level age ~14-16, ~US grades 9-10 Biology revision notes
|
Doc Brown's Biology exam study revision notes: There are various sections to work through, after 1 they can be read and studied in any order. The for a given yeast are: temperature, concentrations of enzyme, concentration of substrate sugar molecules , but yeast produces more consistent results. If you start with sucrose, the enzyme invertase hydrolyses sucrose and breaks it down into glucose and fructose. C H O + H O ===> C H O + C H O The actual anaerobic fermentation reaction is ... glucose/fructose (sugar) == enzyme zymase ==> ethanol + carbon dioxide H O ===> 2C H OH + 2CO You can following the speed of the reaction by measuring the volume of carbon dioxide formed.
You can start at 20 C and repeat the experiments several times for each temperature, and then raise the temperature by 5 C at a time to see the effect. CO /min.Using the above apparatus, or that described below (gas syringe) you can measure the rate of respiration under varying conditions, BUT make sure you only vary one factor, to measure its quantitative effect on the rate of respiration. , same enzyme concentration, same temperature(ii) for a , the effect of at constant temperature and constant enzyme concentration, (iii) for a , the effect of at constant temperature and constant sugar concentration. : you get a white precipitate ('milkyness'), a positive test for carbon dioxide from the yeast anaerobic respiration..You get exactly the same result if you blow some of your expelled breath through limewater - the same carbon dioxide from your aerobic respiration.
Doing the experiments at constant room temperature, you can keep the yeast concentration constant and OR you can vary the substrate sugar (but keeping the sugar concentration constant). of evolution of carbon dioxide e.g. CO /minute.
(gcse chemistry notes) Know the apparatus required from a diagram describe an outline of the procedure. Know how to record the results and from the data graphs to show the variation of the rate of aerobic respiration with temperature or varying the concentration of the sugar (or yeast).
This is a BIG website, so try using the [ ], it maybe quicker than the many indexes! for UK KS3 science students aged ~12-14, ~US grades 6-8* * for UK GCSE level students aged ~14-16, ~US grades 9-10 for pre-university age ~16-18 ~US grades 11-12, K12 Honors Website content � Dr Phil Brown 2000+. All copyrights reserved on Doc Brown's biology revision notes, images, quizzes, worksheets etc. Copying of website material is NOT permitted. Exam revision summaries and references to science course specifications are unofficial. |
| specific biology words or courses e.g. topic, module, exam board, structure, concept, animal or plant feature, 'phrase', homework question! anything of biological interest! This is a Google generated search of my website. |
TOP OF PAGE
Science Bob
- Experiments
- Science Fair Ideas
- Science Q&A
- Research Help
- Experiment Blog
more in Experiments
Blow up a balloon with yeast, you will need.
A packet of yeast (available in the grocery store) A small, clean, clear, plastic soda bottle (16 oz. or smaller) 1 teaspoon of sugar Some warm water A small balloon
1. Fill the bottle up with about one inch of warm water. ( When yeast is cold or dry the micro organisms are resting.) 2. Add all of the yeast packet and gently swirl the bottle a few seconds. (As the yeast dissolves, it becomes active – it comes to life! Don’t bother looking for movement, yeast is a microscopic fungus organism.) 3. Add the sugar and swirl it around some more. Like people, yeast needs energy (food) to be active, so we will give it sugar. Now the yeast is “eating!”
4. Blow up the balloon a few times to stretch it out then place the neck of the balloon over the neck of the bottle. 5. Let the bottle sit in a warm place for about 20 minutes If all goes well the balloon will begin to inflate!
How does it work?
As the yeast eats the sugar, it releases a gas called carbon dioxide. The gas fills the bottle and then fills the balloon as more gas is created. We all know that there are “holes” in bread, but how are they made? The answer sounds a little like the plot of a horror movie. Most breads are made using YEAST. Believe it or not, yeast is actually living microorganisms! When bread is made, the yeast becomes spread out in flour. Each bit of yeast makes tiny gas bubbles and that puts millions of bubbles (holes) in our bread before it gets baked. Naturalist’s note – The yeast used in this experiment are the related species and strains of Saccharomyces cervisiae. (I’m sure you were wondering about that.) Anyway, when the bread gets baked in the oven, the yeast dies and leaves all those bubbles (holes) in the bread. Yum.
MAKE IT AN EXPERIMENT
The project above is a DEMONSTRATION. To make it a true experiment, you can try to answer these questions:
1. Does room temperature affect how much gas is created by the yeast? 2. Does the size of the container affect how much gas is created? 3. What water/room temperature helps the yeast create the most gas? 4. What “yeast food” helps the yeast create the most gas? (try sugar, syrup, honey, etc.)
More Images & Video

More from my site

ADS (these ads support our free website)
Share this page.
Your browser is not supported
Sorry but it looks as if your browser is out of date. To get the best experience using our site we recommend that you upgrade or switch browsers.
Find a solution
- Skip to main content
- Skip to navigation

- Back to parent navigation item
- Primary teacher
- Secondary/FE teacher
- Early career or student teacher
- Higher education
- Curriculum support
- Literacy in science teaching
- Periodic table
- Interactive periodic table
- Climate change and sustainability
- Resources shop
- Collections
- Remote teaching support
- Starters for ten
- Screen experiments
- Assessment for learning
- Microscale chemistry
- Faces of chemistry
- Classic chemistry experiments
- Nuffield practical collection
- Anecdotes for chemistry teachers
- On this day in chemistry
- Global experiments
- PhET interactive simulations
- Chemistry vignettes
- Context and problem based learning
- Journal of the month
- Chemistry and art
- Art analysis
- Pigments and colours
- Ancient art: today's technology
- Psychology and art theory
- Art and archaeology
- Artists as chemists
- The physics of restoration and conservation
- Ancient Egyptian art
- Ancient Greek art
- Ancient Roman art
- Classic chemistry demonstrations
- In search of solutions
- In search of more solutions
- Creative problem-solving in chemistry
- Solar spark
- Chemistry for non-specialists
- Health and safety in higher education
- Analytical chemistry introductions
- Exhibition chemistry
- Introductory maths for higher education
- Commercial skills for chemists
- Kitchen chemistry
- Journals how to guides
- Chemistry in health
- Chemistry in sport
- Chemistry in your cupboard
- Chocolate chemistry
- Adnoddau addysgu cemeg Cymraeg
- The chemistry of fireworks
- Festive chemistry
- Education in Chemistry
- Teach Chemistry
- On-demand online
- Live online
- Selected PD articles
- PD for primary teachers
- PD for secondary teachers
- What we offer
- Chartered Science Teacher (CSciTeach)
- Teacher mentoring
- UK Chemistry Olympiad
- Who can enter?
- How does it work?
- Resources and past papers
- Top of the Bench
- Schools' Analyst
- Regional support
- Education coordinators
- RSC Yusuf Hamied Inspirational Science Programme
- RSC Education News
- Supporting teacher training
- Interest groups

- More navigation items
Yeast and the expansion of bread dough
In association with Nuffield Foundation
- No comments
Try this class practical to investigate how temperature affects yeast and the expansion of bread dough
Yeast is a microbe used in bread making which feeds on sugar. Enzymes in yeast ferment sugar forming carbon dioxide and ethanol. The carbon dioxide makes the bread rise, while the ethanol evaporates when the bread is baked. In this experiment, students investigate the effect of different temperatures on yeast activity and the expansion of the bread dough.
It is best if each group does the activity at one temperature and then shares the results with other groups.
- Spatula or glass rod
- Beaker, 100 cm 3
- Measuring cylinder, 250 cm 3
- Measuring cylinder, 50 cm 3
- Thermometer, 0–100 °C
- Graph paper
- Access to a balance (1 d.p.)
- Access to water baths set at 20 °C, 30 °C and 37 °C (see note 2 below)
- Plain flour, 25 g
- Yeast suspension, 30 cm 3 (see note 3 below)

Health, safety and technical notes
- Read our standard health and safety guidance.
- If thermostatically controlled water baths are not available then large beakers of water maintained at the three temperatures can be used. The water temperature in each beaker needs to be monitored using a thermometer, and access to a supply of hot water, eg from a kettle, is needed to top up the beaker as the temperature falls.
- The yeast suspension is made by stirring 7 g of dried yeast with 450 cm 3 of warm water. If fresh yeast is used it should not have been kept too long. Fresh yeast can be kept in the freezer for up to two months.
- Add 25 g of flour to a beaker and then add 1 g of sugar.
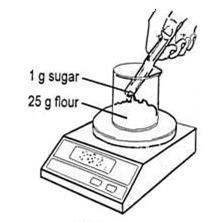
Source: Royal Society of Chemistry
Weigh flour and sugar for a simple bread dough
- Take 30 cm 3 of the yeast suspension in a 50 cm 3 measuring cylinder. Add the yeast suspension to the flour and sugar. Stir with a spatula or glass rod until a smooth paste, which can be poured, has been obtained.
- Pour the paste into a 250 cm 3 measuring cylinder. Take great care not to let the paste touch the sides – this is very important.
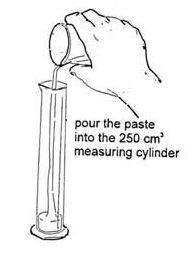
Make a paste from the flour, sugar and yeast suspension and pour it into a measuring cylinder
- Note the volume of paste in the cylinder. Place the cylinder in one of the water baths. Record the temperature and note the volume of paste every two minutes for about 30 minutes. A results table is useful here.
- Plot a graph to show how the volume of the dough increases with time. Plotting the results from groups, with the water baths at different temperatures, on the same graph will allow comparison of results.
Teaching notes
It is important that the paste does not touch the sides of the measuring cylinder when the students pour it from the beaker – this is easier said than done. One way of achieving this is to use a large plastic funnel which has had the stem cut off to leave a hole large enough for the paste to flow through the funnel. Alternatively, a suitable plastic drinks bottle could be cut to produce a wide mouthed ‘funnel’.
After 35–45 minutes the protein (gluten) breaks and carbon dioxide gas escapes.
Possible extensions for this activity could be to investigate the effect of substrate concentration (sugar), enzyme concentration and/or pH value on enzyme reactions.
Additional information
This is a resource from the Practical Chemistry project , developed by the Nuffield Foundation and the Royal Society of Chemistry.
Practical Chemistry activities accompany Practical Physics and Practical Biology .
© Nuffield Foundation and the Royal Society of Chemistry
- 11-14 years
- 14-16 years
- Practical experiments
- Biological chemistry
Specification
- 7. Investigate the effect of a number of variables on the rate of chemical reactions including the production of common gases and biochemical reactions.
- 9. Consider chemical reactions in terms of energy, using the terms exothermic, endothermic and activation energy, and use simple energy profile diagrams to illustrate energy changes.
- 2. Develop and use models to describe the nature of matter; demonstrate how they provide a simple way to to account for the conservation of mass, changes of state, physical change, chemical change, mixtures, and their separation.
- Awareness of the contributions of chemistry to society, e.g. provision of pure water, fuels, metals, medicines, detergents, enzymes, dyes, paints, semiconductors, liquid crystals and alternative materials, such as plastics, and synthetic fibres; increasi…
- Enzymes as catalysts produced by living cells (two examples).
Related articles

Why are some plants poisonous to you and your pets?
2024-05-22T08:16:00Z By Kit Chapman
Dig up the toxic secrets of nature’s blooms

Why fermented foods are good for your gut – and your teaching
2024-04-15T05:30:00Z By Emma Davies
From kimchi to kefir, tuck into the complex chemistry of fermentation and its health potential

Sniffing out the science of smells
2024-03-25T04:00:00Z By Hayley Bennett
What makes a bad smell smell bad? Sniff out the chemical culprits behind obnoxious odours
No comments yet
Only registered users can comment on this article., more experiments.

‘Gold’ coins on a microscale | 14–16 years
By Dorothy Warren and Sandrine Bouchelkia
Practical experiment where learners produce ‘gold’ coins by electroplating a copper coin with zinc, includes follow-up worksheet

Practical potions microscale | 11–14 years
By Kirsty Patterson
Observe chemical changes in this microscale experiment with a spooky twist.

Antibacterial properties of the halogens | 14–18 years
By Kristy Turner
Use this practical to investigate how solutions of the halogens inhibit the growth of bacteria and which is most effective
- Contributors
- Email alerts
Site powered by Webvision Cloud


We welcome your feedback, comments and questions about this site or page. Please submit your feedback or enquiries via our Feedback page.
- International
- Education Jobs
- Schools directory
- Resources Education Jobs Schools directory News Search

Effects of temperature on bread dough
Subject: Design, engineering and technology
Age range: 14-16
Resource type: Worksheet/Activity
Last updated
16 July 2020
- Share through email
- Share through twitter
- Share through linkedin
- Share through facebook
- Share through pinterest

Tes classic free licence
Your rating is required to reflect your happiness.
It's good to leave some feedback.
Something went wrong, please try again later.
Empty reply does not make any sense for the end user
excellent resource. Brilliant for extension work during the Agriculture and Food Applied Science topic. Lost one star as it is more realistic with tight budgets to do an activity based on this using repiration rates of yeast, but excellent to relate to use of yeast in bread making.
Report this resource to let us know if it violates our terms and conditions. Our customer service team will review your report and will be in touch.
Not quite what you were looking for? Search by keyword to find the right resource:

IMAGES
COMMENTS
Investigate anaerobic respiration in yeast with this step-by-step investigation worksheet. With the aim to observe the process of respiration in yeast, this Anaerobic Respiration in Yeast Investigation printout clearly explains the equipment, safety information and method required to complete this investigation. When done correctly, students will have a visual sign that will confirm anaerobic ...
This experiment uses a living organism to investigate the conditions under which life grows the best.(Part 8 of 10)Playlist link - http://www.youtube.com/pla...
This video looks at the uses of anaerobic respiration (fermentation) in yeast and how the ethanol and carbon dioxide that are produced, can be used. Work al...
Swirl the flask to dissolve the glucose. Add 1 g of yeast to the solution and loosely plug the top of the flask with cotton wool. Wait while fermentation takes place. The time it takes will depend on the temperature, how well you mixed the reactants and the yeast's freshness. Add 5 cm 3 of limewater to the boiling tube.
Empty reply does not make any sense for the end user. Submit reply Cancel
Task B is an experiment which students could carry out themselves. Teacher tip. Content guidance. Depiction or discussion of mental health issues ... Correct answer: Laura: Anaerobic cellular respiration is carried out by yeast and bacteria. Laura: Anaerobic cellular respiration is carried out by yeast and bacteria. Alex: Anaerobic cellular ...
Investigate anaerobic respiration in yeast with this step-by-step investigation worksheet. With the aim to observe the process of respiration in yeast, this printout clearly explains the equipment, safety information and method required to complete this investigation. When done correctly, students will have a visual sign that will confirm ...
Anaerobic respiration by yeast - fermentation | Physiology | Biology | FuseSchoolCan you name a type of fungus that is important in the diet of humans? I bet...
yeast suspension (100g/dm 3) Sucrose solution (0.4moldm -3) thermometer. access to hot and cold water. 1dm 3 beaker for carrying water. 20cm 3 syringe. weight. trough. marker pen.
The lesson is in keeping with the current KS3 UK National Curriculum, is easily accessible for students and simple to follow for teachers. Part of this lesson directs students to access the Year 8 Exploring Science textbook; however, it can be equally well performed using any resource that outlines how fermentation occurs in yeast.
Investigating the chemistry of the anaerobic respiration of yeast cells. You can also do the experiment with germinating seeds, but ... Basic Science Quizzes for UK KS3 science students aged ~12-14, ~US grades 6-8. Biology * Chemistry * Physics for UK GCSE level students aged ~14-16, ...
1. Fill the bottle up with about one inch of warm water. ( When yeast is cold or dry the micro organisms are resting.) 2. Add all of the yeast packet and gently swirl the bottle a few seconds. (As the yeast dissolves, it becomes active - it comes to life! Don't bother looking for movement, yeast is a microscopic fungus organism.)
Investigate anaerobic respiration in yeast with this step-by-step investigation worksheet. With the aim to observe the process of respiration in yeast, this printout clearly explains the equipment, safety information and method required to complete this investigation. When done correctly, students will have a visual sign that will confirm anaerobic respiration in yeast and will be able to ...
2-3 Tablespoons lukewarm water. Party balloon. Bowl or mug full of lukewarm water. Inflating a balloon with yeast is a wonderful experiment to do with preschool and kindergarten aged children because all of the materials are nontoxic. It's nice when the kids can help measure out ingredients without worrying about what they are touching.
Take 30 cm 3 of the yeast suspension in a 50 cm 3 measuring cylinder. Add the yeast suspension to the flour and sugar. Stir with a spatula or glass rod until a smooth paste, which can be poured, has been obtained. Pour the paste into a 250 cm 3 measuring cylinder. Take great care not to let the paste touch the sides - this is very important.
Fungi kingdom, Yeast reproduction, conditions for growth rate of a yeast population, Yeast Experiment: measuring respiration in yeast, A series of free Science Lessons for 7th Grade and 8th Grade, KS3 and Checkpoint, GCSE and IGCSE Science, examples and step by step demonstration
Investigate anaerobic respiration in yeast with this step-by-step investigation worksheet. With the aim to observe the process of respiration in yeast, this printout clearly explains the equipment, safety information and method required to complete this investigation. When done correctly, students will have a visual sign that will confirm ...
Age range: 11-14. Resource type: Worksheet/Activity. File previews. ppt, 401.5 KB. ppt, 608 KB. docx, 10.9 KB. doc, 34 KB. Investigation of affect of sugar levels on respiration in yeast. Uses results to discuss how the spread of results around a line of best fit can be used to asses reliability of results.
A funnel. A tablespoon. White sugar. Baking soda. Measuring cups or a jug. Warm water. Six packets of dry yeast. A bowl of warm water or a warm area like a windowsill. Carry out this yeast experiment for preschoolers to observe, or allow older children to carry out the experiment themselves and develop their science skills.
Derived from seaweed, agar is a gelatinlike ingredient used to grow yeast on a solid surface. It's like flour in cake batter, Protacio says. "You'd never expect the flour—it's the most basic thing." And yet here was agar, foiling day after day of experiments. As is turned out, Protacio's lab wasn't alone.
1 INTRODUCTION 1.1 Background and Terms of Reference as provided by the requestor. On 26 March 2021, the company Hayashibara Co., Ltd. 1 submitted a request to the Commission in accordance with Article 10 of Regulation (EU) 2015/2283 to place on the EU market glucosyl hesperidin. Glucosyl hesperidin is intended to be used for addition to several hot beverages, non-alcoholic beverages ...
KS3 Unit 8D Biology - Cells, fungi and the carbon cycle. Aimed for High-Middle ability Year 8 but can be adapted for low/middle ability if required (Higher and lower slides) ... To evaluate the experiment Lesson 4 - Yeast, alcohol production and Growth curves To explain what happens in fermentation
swf, 30.35 KB. Aims Students should: * understand the role yeast plays in bread making * know the effect that temperature and sugar (substrate) concentration have on the rate of bread dough expansion * understand the need to control variables in investigations Contents * Starter (including animation) outlining how yeast causes dough to rise.