

5 Ideas for Physical & Chemical Change: Experiments & Demonstrations They’ll Love

These ideas aren’t your average science experiments! We’re going to dig deeper with conservation of matter, CER, and a phenomenon-based science unit.
There are MANY physical and chemical changes you can observe, but here are 5 of my favorites for upper elementary. Many of these ideas include a focus on conservation of matter because the two concepts go hand in hand.
Physical changes are changes to the appearance or form of a substance, but the substance itself is not changed into a new substance. Chemical changes involve a substance changing into a new substance with different properties.
1. 🥛 Soda Surprise Investigation

This scenario is provided at the beginning of the chemical change unit . Students discuss the scenario then investigate what occurs when soda and milk are mixed. Warning: It’s pretty gross but makes for a meaningful introduction!
Try it on your own or see the unit on TpT: Chemical Change Phenomena-based Science
2. 🎈 Baking Soda & Vinegar Balloon

Take the traditional baking soda and vinegar reaction and add a balloon . When a balloon of baking soda is placed over a graduated cylinder of vinegar in a closed system , you can more easily observe the production of a gas. The reaction occurs and the balloon inflates!
There are many variations of this investigation from simple observation to experimental design to proving the conservation of matter.
This station is part of a stations set on TpT: Physical and Chemical Change Stations
3. 🌟 Glow Sticks

The glow stick is the perfect item to explore chemical change.
It’s cheap.
It’s not messy.
It’s a closed system.
Measure the mass of the glow stick before and after the chemical reaction to demonstrate the conservation of matter.
Try it on your own or see the unit on TpT: Conservation of Matter Phenomena-based Science
4. 🍬 Dissolving Sugar Cubes

Dissolving is a simple observable physical change. I like sugar cubes for ease. In this lab, students demonstrate the conservation of matter while observing a physical change.
- balance or scale
- beaker with 100 ml water
- sugar cubes
- stirring rod/craft stick
- Measure the mass of the cup.
- Add 100 ml of water to the cup.
- Measure 25 g of sugar cubes.
- Add the sugar cubes to water and stir to dissolve.
- Measure the mass of the solution. Be sure to subtract the mass of the cup.
5. 🥤 Mentos + Diet Coke Demonstration

A fun way to wrap up your unit? Do the classic Mentos and Diet Coke demonstration!
Ask students to determine whether a physical change or chemical reaction occurred .
This one is tricky because it certainly seems like a chemical reaction similar to baking soda and vinegar, but it’s actually a physical reaction. Tiny bumps along the candy’s surface make the bonds between water and carbon dioxide easily broken, bubbling up, and resulting in that cool eruption.
Top Teaching Tools
Sign up for the free resource library.
This is an exclusive library of 40+ science printables, labs, activities, and games for grades 3-6. Sign up and check your email for immediate access.
- Read more about: 5th Grade , Physical Science
You might also like...

The Science Penguin Mini PD

CER (Claim Evidence Reasoning) in 3rd, 4th, and 5th Grade Science

5th Grade Science STAAR: 3 Tips to Master Short Constructed Response (SCR)

Hi, I'm Ari!
As a new teacher, I struggled to plan engaging, rigorous science lessons. Throughout my time teaching upper elementary and in my graduate studies, I discovered what worked well and developed science curriculum for busy teachers. Now, teachers across the country use Science Penguin activities every single day in their classrooms!
Want access to The Science Penguin Free Resource Library?
This is an exclusive library of 40+ science printables, labs, activities, and games for grades 3-6! Enter your personal email so your resources don't get stuck in a district filter!
Welcome to Clariclass! If you are interested in signing your child up for our program, please fill out this form!
5th Grade Science
Physical and chemical changes.
Previous Unit
Test & Try
Physical vs. Chemical
Change! It happens all around us every single day, but have you ever stopped to wonder if that change is physical or chemical ? Everything around us can change physically or chemically, and in this unit, we discover how to identify the different changes and take a dip into the world of chemistry .
What's The Matter?
Before we dive into changes, it's important to know and review matter! Matter can be too small to see, but it still exists, and is all around us. Check out these videos to learn more! To test your knowledge, play this game!
https://www.quia.com/hm/40795.html
Physical Change
The key thing to understand when learning about physical changes is that physical changes do not make a new substance . The definition of physical change is a change in the shape or appearance of an object, like crumpling a piece of paper, or cutting, bending, or dissolving something. Physical changes also happen when matter changes states.
Check out the video to your right to get a deeper understanding of physical changes!
Chemical Change
With chemical changes, a key thing to remember is that chemical changes make new substances . Anytime a new substance is made, a chemical change takes place. Usually two or more materials are combined and a new substance is formed.
Check out the video to your left to get a deeper understanding of chemical changes!
How Do We Compare Them?
Alright, now that we know what physical and chemical changes are separately, let's compare them. Start with this video, which gives you a great idea of how we compare the two types of changes.
https://www.flocabulary.com/unit/physical-chemical-changes/
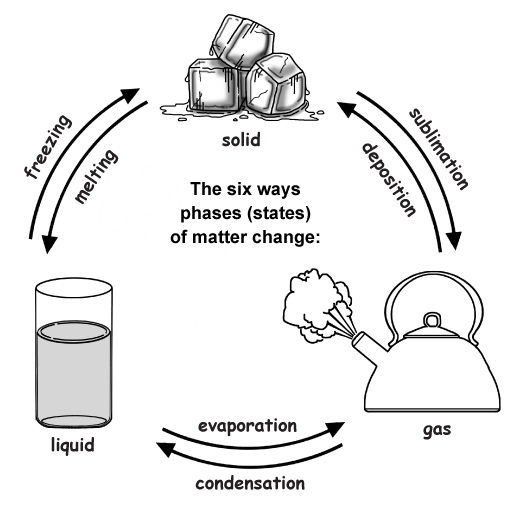
When a solid turns into a liquid it is called melting . The temperature at which this happens called the melting point . As the energy in the molecules increases from a rise in temperature, the molecules start moving faster. Then, the matter becomes a liquid! It's like magic -- or science.
When the opposite happens and a liquid turns into a solid, it is called freezing . The temperature at which this happens called the freezing point . The energy in the molecules decreases from a drop in temperature and the molecules start to slow down, clumping up together. You see this happen when you freeze water!
Do You Think This is a Physical Change or a Chemical Change?
Correct Answer : Freezing and melting are both physical changes! Do you know why? Try to think about this one on your own and keep it in mind for future discussions.
Hint: The answer may be somewhere on this page!
Time for some labs! Check these out, and make sure to watch them all the way. They have very important information that is very useful in understanding this unit!
Feel Confident About This?
Check this game out! It's a jeopardy game for physical and chemical changes. Make sure you've watched all videos and reviewed this page thoroughly so you can do well on this game!
https://jeopardylabs.com/play/physical-and-chemical-properties-and-changes
Think you got it all down? Try this Quizizz! Remember, try as many times until you get your highest score, and come back to this page if you feel unsure about any topic.

Physical and Chemical Changes Activities for 5th Grade
Are you looking for fun and easy physical and chemical changes activities for 5th grade? You can do these experiments at home or in the classroom. I will also show you some great sketch notes and worksheets covering matter, properties of matter, and physical and chemical changes.
Affiliate links for Amazon are included.
Activities for Physical Change
1. skittles in water: sugar dissolving.
Simply arrange the Skittles on a paper plate, add water, and watch as the colored sugar coating dissolves. Here is a link to get Skittles on Amazon. I like to have items delivered right to school.

2. Chocolate Melting
This is a great idea that my partner teacher suggested. Melt white chocolate, dark chocolate, and milk chocolate in the microwave. But before you start, have each student make a hypothesis about which will melt first and which will melt last. Change the color of the plate from white to black and see what happens. Then, eat some chocolate! Amazon link to chocolate here .
Here are some slides to help with a melting chocolate experiment:
https://www.lcps.org/cms/lib/VA01000195/Centricity/Domain/5507/Science_Investigations_example.pptx

3. Shaping Playdough
Students can use their hands, cookie cutters, or molds to shape the dough, It is still play dough so it is a physical change with a change in shape. Amazon link to little individual Play Doh tubs here.

4. Cutting Paper Snow Flakes or Papel Picado
See the video attached or find another student friendly activity on YouTube.

Activities for Chemical Change
1. elephant toothpaste.

2. Diet Coke and Mentos

| I highly recommend the geyser tube from Amazon. |
3. Baggie Science - Hot and...
4. baggie science - cold.
| to see these baggie experiments in my TPT store |
5. Bake Cookies
Any type of baking is a chemical change. This might be great to have students bake at home with an adult and explain the chemical change on SeeSaw.

Are you looking for notes for teaching physical and chemical changes? These notes also include properties of matter.
| to see on TPT |
This resource works towards covering NGSS 5PS1-3 and 5PS1-4. This physical and chemical changes resource includes:
- 8 pages of visual notes
- a slideshow to guide students as they complete the notes
- video link to YouTube
- a link to a Kahoot and a Quizizz game
- CER options
- a CER rubric
- a printable and self-grading digital quiz

Get Your ALL ACCESS Shop Pass here →

Examples Of Physical Change
What is a physical change? Learn to identify a physical change vs. a chemical change with a simple physical change definition and everyday examples of physical change. Explore physical changes with easy, hands-on science experiments kids will love. Fun science project ideas for all ages of kids!

What Is A Physical Change?
Physical changes are changes that occur in matter without changing its chemical composition. In other words, the atoms and molecules that make up the matter stay the same; no new substance is formed . But there is a change in the appearance or physical properties of the substance.
Physical properties include:
- Temperature
For Example…
Crushing an aluminum can: The aluminum can is still made of the same atoms and molecules, but its size has changed.
Tearing paper: The paper is still made of the same atoms and molecules, but its size and shape have changed.
Freezing water: When water freezes, its appearance changes from a liquid to a solid, but its chemical composition remains the same.
Dissolving sugar in water: The sugar and water are still made of the same atoms and molecules, but their appearance has changed.
Understanding physical changes is important for many fields, such as physics , engineering , and materials science. It helps us understand how matter behaves and how to manipulate it.
Physical vs. Chemical Change
Physical changes differ from chemical changes or chemical reactions, which occur when the substances are changed into one or more new substances. A chemical change is a change in the chemical composition of the matter. In contrast, a physical change isn’t! Learn more about chemical change.
For example, when wood burns, it undergoes a chemical change and turns into a different substance, ash, which has different atoms and molecules from the original wood.
However, if a piece of wood is chopped up into smaller pieces, it undergoes a physical change. The wood looks different, but has the same substance as the original wood. Learn more about physical change.
💡 SUGGESTION: Fun Chemical Reaction Experiments
Physical changes are often reversible, especially if they are phase changes. Examples of phase changes are melting (changing from a solid to a liquid), freezing (changing from a liquid to a solid), evaporation (changing from a liquid to a gas), and condensation (changing from a gas to a liquid).
A great question for kids to ask is… Can this change be reversed or not?
Many physical changes are reversible . However, some physical changes are not easy to reverse! Think about what happens when you shred a piece of paper! While you haven’t created a new substance, the change is irreversible. Chemical changes typically are irreversible .
Everyday Examples Of Physical Change
Here are 20 everyday physical change examples. Can you think of any more?
- Boiling a cup of water
- Adding milk to cereal
- Boiling pasta to make it soft
- Munching on candy
- Chopping vegetables into smaller pieces
- Grating an apple
- Melting cheese
- Slicing a loaf of bread
- Washing clothes
- Sharpening a pencil
- Using an eraser
- Crushing a box to put in the trash
- Steam condensing on the mirror from a hot shower
- Ice on the car window on a cold morning
- Mowing the lawn
- Drying clothing in the sun
- A puddle of water drying up
- Trimming trees
- Adding salt to a pool
Grab this FREE Physical Change Info pack to get started!

Physical Change Experiments
Try one or more of these easy physical change experiments you can do at home or in the classroom. What physical changes can you observe? For some of these experiments, there may be more than one.
Crushed Can Experiment
Observe how changes in atmospheric pressure can crush a can. A fun and easy experiment to try!

Dissolving Candy
Add candy to water for a fun, colorful physical change. Also, investigate what happens when you add candy to other common household liquids.
Freezing Water Experiment
Learn about the freezing point of water and what kind of physical change happens when you add salt to water and freeze it.

Ivory Soap Experiment
What happens to ivory soap when you heat it in the microwave? Observe a cool physical change in action!
Making Paper
Make these paper Earths from old bits of paper. The appearance of the paper changes with this easy recycling paper project.
Making Soap
Not only is making soap from a melt and pour glycerin base a fun example of physical change, you also end up with a cool surprise at the end.
Melting Ice Experiment
What makes ice melt faster? 3 fun experiments to investigate what speeds up the process of ice changing from a solid to a liquid.

Melting Crayons
Turn a box of broken and worn down bits of crayon into new crayons with a fun example of physical change. Follow our step by step instructions to melt the crayons, and make them into new crayons.
Paper Towel Art
What kind of physical change do you get when you add water and ink to a paper towel? This also makes for a fun and easy STEAM (Science + Art) activity.
For another “arty” example of physical change, try salt painting !
Popcorn In A Bag
Science you can eat! Make up some popcorn in a bag, and find out what type of physical change makes popcorn pop.

Rainbow In A Jar
How does adding sugar to water cause a physical change? It changes the density of the liquid. See it in action with this colorful layered density tower.
Salt Water Density Experiment
Similarly, explore how adding salt to water changes the physical properties of the water. Test it out by floating an egg.

Skittles Experiment
Use your skittles candy and water for this classic skittles science experiment that everyone has to try! Why don’t the skittles colors mix?
Solid, Liquid, Gas Experiment
A simple science experiment that is great for younger kiddos. Observe how ice becomes a liquid and then a gas.
What Absorbs Water
A simple experiment for your preschoolers! Grab some materials and objects, and investigate what absorbs water and what doesn’t. Physical changes you may notice; changes in volume, texture (wet or dry), size, color.
Compare how fast different everyday items melt in the sun, including ice cubes. A fun experiment to do in the summer!
Physical Change Experiments That Look Like Chemical Reactions
The science experiments below are all examples of physical change. While, at first, you might think a chemical reaction has occurred, all that fizzing action is a physical change!
Dancing Raisins
While it might appear that a chemical change is occurring, a new substance is not formed. The carbon dioxide, which is found in the soda, creates the movement of the raisins.

Diet Coke and Mentos
Adding Mentos candy to Diet coke or soda makes for the best explosion! It is all to do with a physical change! Check out our Mentos and soda version for younger kids as well.

Pop Rocks and Soda
Mix Pop rocks and soda together for a frothy, fizzing physical change that can blow up a balloon.

More Helpful Science Resources
Here are a few resources to help you introduce science more effectively to your kiddos or students and feel confident when presenting materials. You’ll find helpful free printables throughout.
- Best Science Practices (as it relates to the scientific method)
- Science Vocab
- 8 Science Books for Kids
- What Is A Scientist
- Science Supplies List
- Science Tools for Kids
Science Experiments By Age Group
We’ve put together a few separate resources for different age groups, but remember that many experiments will cross over and can be re-tried at several different age levels. Younger kiddos can enjoy the simplicity and hands-on fun. At the same time, you can talk back and forth about what is happening.
As kiddos get older, they can bring more complexity to the experiments, including using the scientific method , developing hypotheses, exploring variables , creating different tests, and writing conclusions from analyzing data.
- Science for Toddlers
- Science for Preschoolers
- Science for Kindergarten
- Science for Early Elementary Grades
- Science for 3rd Grade
- Science for Middle School
Printable Science Experiments Pack
If you’re looking to grab all of our printable science projects in one convenient place plus exclusive worksheets and bonuses like a STEAM Project pack, our Science Project Pack is what you need! Over 300+ Pages!
- 90+ classic science activities with journal pages, supply lists, set up and process, and science information. NEW! Activity-specific observation pages!
- Best science practices posters and our original science method process folders for extra alternatives!
- Be a Collector activities pack introduces kids to the world of making collections through the eyes of a scientist. What will they collect first?
- Know the Words Science vocabulary pack includes flashcards, crosswords, and word searches that illuminate keywords in the experiments!
- My science journal writing prompts explore what it means to be a scientist!!
- Bonus STEAM Project Pack: Art meets science with doable projects!
- Bonus Quick Grab Packs for Biology, Earth Science, Chemistry, and Physics

Subscribe to receive a free 5-Day STEM Challenge Guide
~ projects to try now ~.

Rosie Riveters

Rosie Explores Chemical and Physical Change
Perform these fun experiments to explore chemical and physical change!
Homepage • Hands-on STEM • Resources
- Fire is used as a part of these experiments. Please do not perform without an adult.
Learn the differences between chemical and physical change in this series of experiments . We’re playing with paper towels, blowing up balloons, and making all kinds of chemical reactions!
Related Activities

-->Rosie Explores Magnetism -->
Save Resource

-->Rosie Explores Centripetal Force -->

-->Rosie Makes A House That Code Built -->
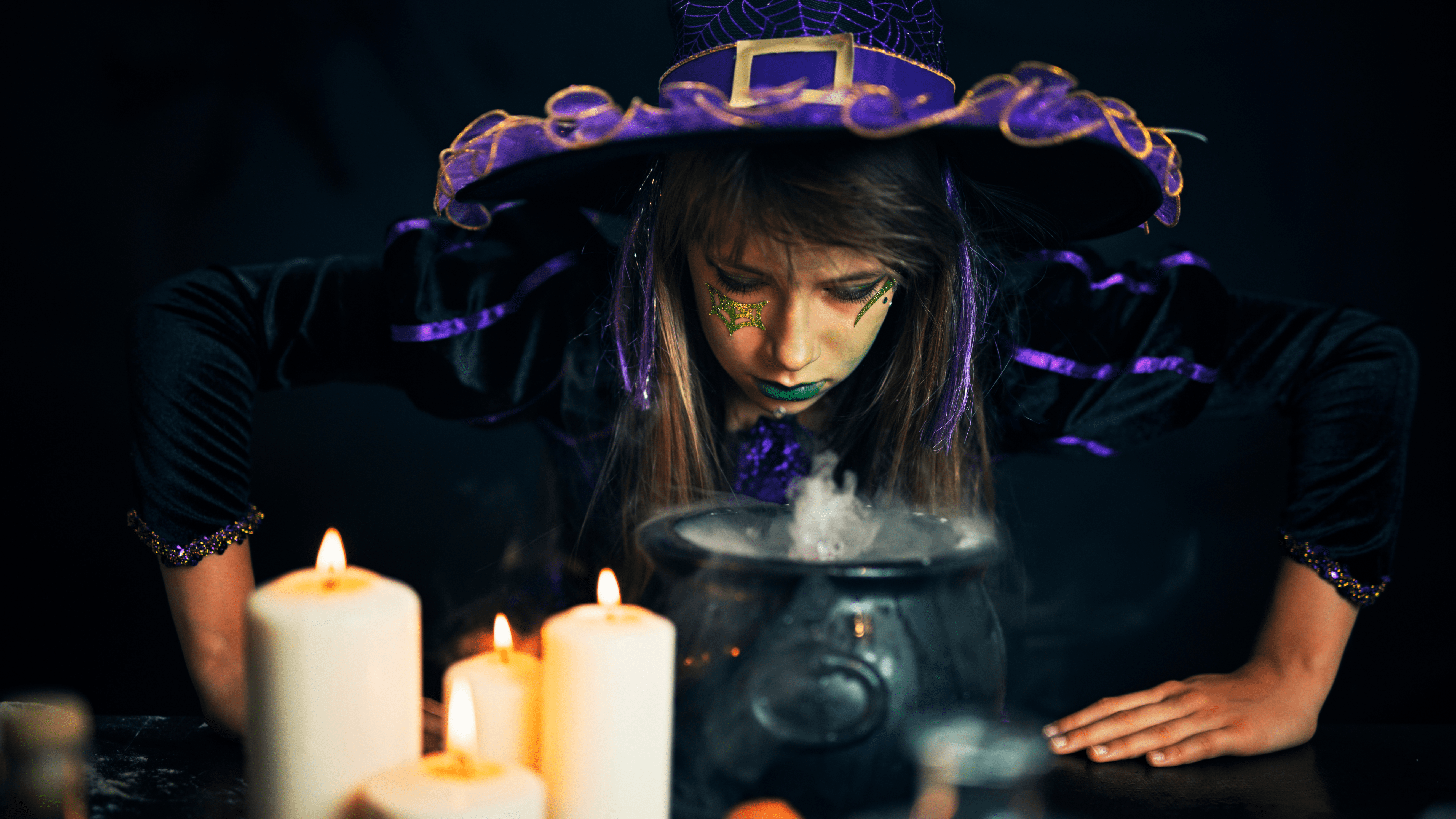
-->Rosie Makes Foaming Witch’s Brew – No Peroxide -->
Get the updates, you found it, create a lesson plan to save your cache.
Create an account and a lesson plan called “Women’s History Hunt” to save this resource.
Once you’ve found and saved all five caches to your lesson plan submit a screenshot to [email protected]!
" * " indicates required fields
Already have an account? Sign In .
Disclaimer related to signing up and agreeing to terms and services and the privacy policy . This can be removed if not need.
Sign Up and Start Saving Resources
Explore hundreds of STEM resources and save them to lesson plan so you can return back them later.
Create a New Lesson Plan
Create lesson plans to save resources for later. Lesson plans are private for your account only.
Welcome Back
Sign in to your account to continue saving resources.
Don’t have an account? Sign Up .

Teaching a Physical and Chemical Change Unit

Are you frustrated with finding ways to get your students to understand how matter changes ? Identifying differences between chemical and physical changes can frustrate the teacher and the student. When I first began teaching, this was a unit I dreaded. I felt intimidated about accurately teaching this concept to my students and didn’t feel confident in myself.
Flash forward over ELEVEN YEARS, and I can now say this is one of my favorite teaching units. While teaching is still challenging, you’ll feel like a rockstar when students have grasped this difficult concept. Have that same rockstar feeling by learning tips and strategies to teach your students a physical and chemical changes unit.
What is a change in matter?
It is essential to know the difference between the two ways that matter can change.

When teaching about color changes to identify a chemical change, you need to differentiate between adding colors such as food coloring or color being produced, like when leaves change color in the fall . Leaves no longer receive what they need, which is why they change color. Mold is another example of a chemical change. Part of the bread changes color because of something new on the bread, the mold. This is important to allow students to understand.
Referencing physical and chemical changes
Aside from anchor charts displayed around my classroom, we also read science texts and go over numerous examples to identify the differences between the two changes. Again, repetition and review are necessary to help students remember and apply their learning.

Hands on Learning

Making claims
Hands-on experiments.
- Elephant’s Toothpaste
- Race of the Ice Cubes
- Air balloons – baking soda and vinegar using a balloon (as seen in the picture below)
- Rates of rotting apples
- Digestion in a bag
- Tarnished coins
- Pumpkin Science

Reviewing Physical and Chemical Changes

Graphic Organizers :
Real-life scenarios :.

Assessments:

I hope these tips help you in your classroom. This unit can be confusing at first glance , but I hope these strategies and ideas will help make it much clearer.
Still have questions? Let me know by leaving them in the comments below.
Looking for some materials to teach this unit, join my email list to help get you started!
- Read more about: blended learning , free , fun , hands-on , physical science , professional development , science , teacher tips
You might also like...

5 Engaging Biome Activities for Grades 5-8

How to Motivate Your Students: Tips for Teachers

5 Ways to Teach about Wildlife Conservation
On instagram, @ateachingmuse.
Please go to the Instagram Feed settings page to create a feed.
Join the Science Squad
- Skip to primary navigation
- Skip to main content
- Skip to primary sidebar
Teaching Expertise
- Classroom Ideas
- Teacher’s Life
- Deals & Shopping
- Privacy Policy
Physical And Chemical Changes Activities For Middle School: Discussions, Experiments, Observation, And Other Resources
January 2, 2024 // by Elizabeth Weinick
Let’s face it – distinguishing between physical and chemical changes is not as easy as differentiating between cutting paper versus an exploding model volcano. There are many misconceptions that make the two concepts difficult for learners to understand! Middle schoolers must have a solid understanding of these concepts in order to apply their knowledge to bigger concepts in later grades. Here are 27 easy-prep, memorable and hands-on activities for middle schoolers to defy their misconceptions about physical and chemical changes and master the basics!
1. An Introduction to Physical and Chemical Changes
This introduction to physical and chemical changes includes a video, discussion questions, a vocabulary review, an activity guide, and assessments. The video is engaging and relatable for middle schoolers!
Learn More: Generation Genius
2. Skittles Science
You can dissolve skittles to investigate the question at the end of the rainbow – is this a physical or chemical change? You can vary the experiment by using different temperatures of water, white vinegar, or even lemonade to discover what happens.
Learn More: Science Sparks
3. Baking Chemistry
Explore chemical changes with baking! Sabrina talks about changes that can’t be undone in this episode of Crash Course Kids. She clearly defines chemical changes and provides a yummy experiment to enjoy after learning!
Learn More: Crash Course Kids
4. Naked Egg
Egg-xamine chemical and physical changes with eggs! This website provides several ways to observe the swelling and shrinking of de-shelled eggs in different liquids. This is also a great option to practice measuring and calculation skills while learning how different liquids affect the eggs’ mass.
Learn More: Exploratorium
5. Maximize Science!
Max captures young scientists’ attention in this video by creating huge versions of typical science experiments. Max investigates the chemical reaction that occurs to create light in a glow stick and the physical change for rock candy before creating a giant version of each!
Learn More: Science Max
6. Tarnished Coins
If you are curious why the Statue of Liberty is green, this activity explains the chemical reaction that happened over time to cause the color to change. This experiment model this oxidation with pennies.
Learn More: wikiHow

7. Dissolving Cups
While you may think watching a styrofoam cup disappear before your eyes is a chemical change, it is actually a physical change! Your children will be amazed to watch the bubbles that appear and learn why this is actually a physical change.
Learn More: Resource Center
8. Elephant Toothpaste
You can create toothpaste that would be perfect for an elephant! The product of this chemical reaction is a large foamy mess that is fun and kid-safe. You may want to dive deeper into why hydrogen peroxide produces this fun substance.
Learn More: Imagination Station Toledo
9. Erupting Diet Coke and Mentos
You know your middle schools are itching for an explosion! Drop mentos into diet coke and squeal while everyone runs to a safe distance to watch the eruption. You can dismantle the misconception that an explosion always means a chemical reaction.
Learn More: Science B uddies
10. CSI Lab
You can bring a CSI television experience into your home with this “Case of the Missing Cake” activity! You and your children must use knowledge of chemical and physical changes to test samples of known and unknown substances to determine which suspect is the culprit!
Learn More: Mrs. Willyerd’s Virtual Classroom
11. Lemon Science
You can make a smaller version of the classic volcano by using a lemon! You may enjoy the low prep and easy-to-find materials needed. The best part is you can see the reaction occurring on the top of the lemon!
Learn More: Learning Lessons with Mrs. Labrasciano
12. Sort it Out
While experiments are a great visual, students also need concrete activities to solidify their knowledge of vocabulary and definitions. Here is a card sort to identify the differences between changes with both words and pictures to help struggling learners.
Learn More: Teaching with Elly Thorsen
13. Create Glow Sticks
We are always amazed at after-dark events with glow sticks! Students will love learning what chemicals must mix to produce the light that entrances us and why that “crack” is important for the change.
Learn More: A Dab Of Glue Will Do
14. Stained Shirt
If you have ever ruined a shirt with bleach, this experiment explains why bleach so powerfully takes over! This experiment is a great hands-on, team project to get all learners involved.
Learn More: Super Sass and Science Class
15. Misconceptions Explained
For struggling learners, this animation breaks down the complex language involved in chemical and physical reactions through an interaction between a scientist and a bunsen burner. They identify many misconceptions, so you may want to discuss with your students which misunderstandings they were surprised about!
Learn More: Smithsonian Science Education Center
16. Air Balloons
Here is a twist on the classic chemical reaction that occurs when you mix baking soda and vinegar! Add a balloon to the top of the container and watch in amazement. You can find out why the chemical reaction causes the balloon to react in this way.
Learn More: Education.com
17. Browning Apples
This experiment proves that we see chemical reactions so often and do not even realize it! Students will explore why the enzymes in apples react with oxygen- and how to prevent this!
Learn More: Purdue
18. Digestion in a Bag
This step-by-step activity puts the knowledge of chemical reactions to use as students investigate how our bodies break down food into small parts to create energy for our bodies. Students will create a model stomach in a ziplock bag!
Learn More: Discovery @ Home
19. Chemical Changes in Everyday Materials
Scientist Jared explains how chemical reactions can happen quickly or slowly. He does this with simple everyday materials, such as tin and fire, and ingredients to make bread.
Learn More: Fun Science Demos
20. Pumpkin Science
Perfect for a fall activity, this experiment allows students to follow the decomposition cycle of a pumpkin and discover what physical and chemical changes are occurring. You may want to supplement this experiment with the books provided!
Learn More: Teaching Muse
21. Popcorn is Physical
You can incorporate chemical and physical reaction learning into snack time! Learners often think physical changes are reversible, however, popcorn is the perfect example of a physical change that we cannot return to the original kernel state. Discuss as you make this snack!
Learn More: Edventures with Kids
22. Get the Scoop on Dairy Products
This lesson allows higher learners to apply their knowledge of physical science to determine what type of changes occur when turning milk into cheese, butter, yogurt, ice cream, whip cream, and other dairy products.
Learn More: Agriculture in the Classroom
23. Make Plastic from Milk
Learn how chemical changes can create something new and usable! Learners can create their own own toys, beads, and more by getting different substances to react with milk. Read on to find out the chemistry and history behind this process!
Learn More: STEAM Powered Family
24. Explore Physical and Chemical Changes in Everyday Life
We don’t always see the explosive attractions that come with chemical and physical reactions. Students can understand that they are seeing these changes in their daily life with stations such as salt and water mixtures, rusted nails, and a browned banana.
Learn More: Bright in the Middle
25. Physical and Chemical Weathering Activities
This project applies concepts of chemical and physical changes to another science topic- weathering! Students can complete these activities with sugar cubes and graham crackers to explore why statues are weathered and why sinkholes occur.
Learn More: The Owl Teacher
26. Kool-Aid Chemical Reactions
While making typical kool-aid is a physical change, complete this experiment with different types of liquids to see how the substance reacts! You may choose between lemon juice, apple cider vinegar, and kool-aid water to see the changes that occur.
Learn More: Teaching Science with Lynda R. Williams
27. Kitchen Science
You can teach physical and chemical properties while baking! You may choose to discuss why common baking ingredients have certain properties, then, enjoy the tasty reward at the end!
Learn More: iExplore Science
Changes in Matter: Physical vs. Chemical Changes
Physical changes do not produce a new substance. Chemical changes result in the production of a new substance and cannot be reversed.
Oxidized Copper Lion
The process of rusting, or oxidization, exemplifies a chemical reaction. Here is an oxidized copper lion statute in front of the Chicago Art Institute and the Aon Center.
Photograph by Paul Damien

Matter is anything that has mass and takes up space. The tiny particles called atoms are the basic building blocks of all matter. Atoms can be combined with other atoms to form molecules . Matter is capable of undergoing changes, which are classified as either physical or chemical. Physical changes in matter are often reversible. For example, an ice cube can melt into liquid water, and then the liquid water can be refrozen into an ice cube. A chemical change is very different. A burning log demonstrates this. The log turns to ashes. However, its ashes cannot be changed back into a log. The burning was a chemical change. Unlike many physical changes, it cannot be easily reversed. What Is a Physical Change? In a physical change, the structure of the changed material remains the same before and after the change. Physical changes may affect the texture, shape, and temperature of a substance. They can also affect the material's state of matter. A change in the texture of a substance is a change in the way it feels. For example, a block of wood may feel rough when you run your finger across it. Rubbing the wood with sandpaper smooths the surface, so it no longer feels rough. This is an example of a physical change: The texture of the wood has changed, but the wood's molecular structure has not. A physical change may also involve a change of phase between solid, liquid, and gas. When a material changes phase, it only changes physically; the substance is still the same, even though its form is different. Think about ice melting into water. It can then be heated up and turned into steam. The phase changes, but the structure of water does not. The water molecules are still water molecules, whether the water is in liquid form, in solid form as ice, or in gas form as steam. What Is a Chemical Change? Chemical changes are very different from physical changes. A chemical change takes place when the composit ion of a substance is altered. This requires the breaking and forming of chemical bonds during a reaction. Chemical bonds hold molecules together. A chemical change results in the rearranging of atoms in substances. This rearrangement leads to the creation of brand new molecules. The new molecules are the products of the chemical reaction . They cannot be easily reverted back to their original state. Sometimes it is not easy to tell whether a chemical reaction has taken place. To figure it out, scientists look for a few basic signs. Indicators of a chemical change can include a change in temperature, color, or odor. In a chemical change, temperature may rise or fall. This release of heat is the result of the breaking or formation of chemical bonds. Burning wood is an example of a reaction that releases energy as heat. The opposite may also indicate a chemical reaction. A chemical change can cause heat to be absorbed. The result is a decrease in temperature. A chemical cold pack in a first aid kit demonstrates this kind of chemical reaction. It absorbs heat energy, which results in cooling. A color change can also signal a chemical reaction. A common example can be observed when you leave an iron nail outside. The iron reacts with oxygen in the air. The reaction produces iron oxide. As a result, the nail develops a reddish-brown rust. The odor of rotting food also indicates a chemical change. The food undergoes chemical reactions as it begins to break down. These changes lead to the formation of new substances. These substances produce unique, often, unpleasant smells. Another sign of a chemical reaction may be the formation of a precipitate . When certain chemicals are mixed together in a solution, a new solid can form in the liquid mixture. This solid is a precipitate. It shows that a chemical reaction has taken place. The change has altered the original substances. A chemical reaction may also release a gas. For example, a mixture of vinegar and baking soda will immediately start bubbling and foaming. The bubbles are a release of carbon dioxide gas, a product of the chemical reaction between the two common substances. Chemical Change or Physical Change? Sometimes it is difficult to tell whether a change is physical or chemical. The formation of alloys is one example. An alloy is a metal made from a mixture of other metals. The alloy's properties are different from the properties of the metals used to make it. Brass is a common example of an alloy. It's used to make musical instruments including trumpets and trombones. Brass is made up of about 60 percent copper and 40 percent zinc. It has different properties than either copper or zinc alone. Yet it is not the result of chemical reaction. That may seem confusing, because brass is different from both copper and zinc. However, though the copper and zinc atoms are both present in brass, they do not chemically bond together. No chemical bonds are formed or broken. As a result, the creation of brass represents a physical change, not a chemical one.
Instructional Links
Media credits.
The audio, illustrations, photos, and videos are credited beneath the media asset, except for promotional images, which generally link to another page that contains the media credit. The Rights Holder for media is the person or group credited.
Production Managers
Program specialists, last updated.
October 19, 2023
User Permissions
For information on user permissions, please read our Terms of Service. If you have questions about how to cite anything on our website in your project or classroom presentation, please contact your teacher. They will best know the preferred format. When you reach out to them, you will need the page title, URL, and the date you accessed the resource.
If a media asset is downloadable, a download button appears in the corner of the media viewer. If no button appears, you cannot download or save the media.
Text on this page is printable and can be used according to our Terms of Service .
Interactives
Any interactives on this page can only be played while you are visiting our website. You cannot download interactives.
Related Resources

Evidence of Chemical Change Labs – Experiments for Kids
These chemical change labs explore five types of evidence: bubbles, color, heat, odor, and light. First, kids experiment with physical change. Then they launch into reactions that create new substances.

Mr. Grow Plans His Physical and Chemical Change Unit
Our favorite fifth grade teacher sat at the side table with his teaching partner. “Today,” he said, “we’ll continue planning our matter activities . This week, our kids are getting an introduction to matter . Next up, physical and chemical change.” He pulled up the standard on his computer and read it aloud:
NGSS 5-PS1-4 Conduct an investigation to determine whether the mixing of two or more substances results in new substances.
“Actually,” said Mrs. Washington, “I’ve been giving this one some thought. Obviously, kids must discriminate between physical and chemical changes. But how? So I did a little research. As it turns out, reactions provide evidence. Through my investigation, I found five types: bubbles, color, heat, odor, and light.”
“Hmm,” Mr. Grow said, “Can kids do an experiment for each one?”
Mrs. Washington’s eyes twinkled. “I already found some physical and chemical change activities .” She beckoned to Mr. Grow, and he moved around to look at her laptop screen.
“Six labs illustrate specific changes. They seem wonderful to me. However, I’d like to try them out. Let’s split them up. Tomorrow we’ll meet again. Then we can try each lab ourselves.”
Physical Change
As promised, the two teachers met the following day. In their arms, each carried a bag of materials. After walking to the side table, they organized everything. Finally, they were ready to share.
“First,” said Mrs. Washington, “let’s try the lab on physical changes.
“For states of matter, they’ll just observe an ice cube. To explore mixtures, they’ll stir together two groups of small items. For example, lima beans and marbles. Then for a solution, they can stir together sugar and water. For all of these activities, kids will work in their science lab groups .”
“How will they respond?”
“For every activity in this sequence, kids fill out a lab sheet. In each, they discriminate between physical and chemical change. You know, was a new substance created? To prove it, they explain the evidence. And we’ll be giving them plenty of it!”

Chemical Change with Bubbles as Evidence
After Mrs. Washington finished, Mr. Grow pointed to his first set-up. “In this lab, kids experiment with vinegar and baking soda.”
Then he demonstrated. With an eyedropper, he added just a few drops of vinegar to the white powder. Soon, bubbles fizzed in the cup. “Here, kids see the first type of evidence of chemical change. If they look carefully, they can also see that the baking soda has changed. A new substance has been created.”
Mrs Washington nodded.

With Color Evidence
Mr. Grow continued. “I wasn’t sure the next experiment would work.”
As he spoke, he added red food coloring to three cups of water. Then he added different liquids to each one.
“First, I’ll add about 20 drops of vinegar to this cup.” Although Mrs. Washington watched carefully, nothing happened.
“Second, I’ll add 20 drops of hydrogen peroxide to this one.” Again, nothing happened.
“Third, I’ll put 20 drops of bleach in the last cup.” At first, nothing happened.
Then – “Oh wow!” Mrs. Washington exclaimed. “The color is fading! For some reason, I expected one of the cups to turn blue or something.”
Mr. Grow laughed. “No, in this lab, evidence of chemical change is color. But here, kids observe loss of it.”
“This is a good one,” Mrs. Washington said. “However, I don’t trust kids to work with bleach. Therefore, we’ll do this as a demonstration.”

With Heat Evidence
“Wait until you see this experiment,” Mr. Grow said. Quickly, he moved to his third lab set-up.
“First, I fill this cup about one-fourth of the way with hydrogen peroxide. Second, I record the temperature.”
He put a thermometer into the liquid and waited a few seconds. “About 71 degrees Fahrenheit.”
“Next, I add a teaspoon of yeast.” Immediately, it began to bubble.
The two teachers stared at the thermometer. “75.3, 78.2, 80!” Over the next few minutes they watched as the temperature topped out at 101.4 degrees.
Mrs Washington placed her hand on the outside of the cup. “Yep, it’s warm. And wow! That was some dramatic demonstration! The kids will love it!”
“This time,” said Mr. Grow, “evidence of chemical change included both bubbles and heat.”

With Odor Evidence
“Now it’s my turn again,” said Mrs. Washington.
She held a cup near Mr. Grow’s nose. “Give it a sniff,” she said.
“Hey, what’s in there?”
“Just some iron filings. Do you smell anything?”
Mr. Grow shook his head. “No.”
Next, Mrs. Washington held up a small container. “Smell again. This is hydrogen peroxide.”
Again, Mr. Grow shook his head. “No strong odor.”
Finally, Mrs. Washington placed a few drops of the liquid in the cup. Quickly, she held the cup near Mr. Grow’s nose.
“Ewww! Rotten eggs!”
Mrs. Washington grinned. Just what she was looking for. A dramatic way to illustrate that odor is evidence of chemical change.

With Light Evidence
Next, Mrs. Washington took out a container of glow sticks.
“The kids will love those!” said Mr. Grow.
Mrs. Washington nodded and smiled. Then she picked up a glow stick and bent it. Snap! Immediately, it began to glow.
“We can explain that the stick is filled with a liquid. Then, bending breaks tiny capsules full of another liquid. As soon as the two meet, they form a new substance, and light energy is emitted.”
“Perfect!”

Enjoy Teaching Physical and Chemical Change Labs
Both teachers grinned.
“You know, these chemical change labs will be a hit,” said Mr. Grow. “Next, we’ll work on the law of conservation of mass and properties of matter .”

Filter Results
- clear all filters
Resource Type
- Guided Lessons
- Lesson Plans
- Hands-on Activities
- Interactive Stories
- Online Exercises
- Printable Workbooks
- Science Projects
- Song Videos
middle-school
- Fine arts
- Foreign language
- Math
- Reading & Writing
- Life Science
- Earth & Space Science
- Engineering & Science Practices
- Social emotional
- Social studies
- Arts & crafts
- Holidays
- Recipes
- Seasonal
Fifth Grade Physical Science Activities


- Next Generation Science Standards
- Grade 5 Science: Proposed by NGSS
Grade 5 - 5-PS1 Matter and Its Interactions
To view these resources with no ads, please login or subscribe to help support our content development. school subscriptions can access more than 175 downloadable unit bundles in our store for free (a value of $1,500). district subscriptions provide huge group discounts for their schools. email for a quote: [email protected] ..
Physical Science F or 5th Grade
LINKS to content and activities to help fulfill the Next Generation Science Standards shown below.
PS1.A: Structure and Properties of Matter
PS1.B: Chemical Reactions
Phases of Matter – Gas, Liquids, Solids
Phases of Matter Matching
Next Generation of Science Standards (NGSS) - Grade 5
Disciplinary Core Ideas
Science and Engineering Practices (NGSS)
Developing and Using Models Modeling in 3–5 builds on K–2 experiences and progresses to building and revising simple models and using models to represent events and design solutions. • Use models to describe phenomena. (5-PS1-1)
Planning and Carrying Out Investigations Planning and carrying out investigations to answer questions or test solutions to problems in 3–5 builds on K–2 experiences and progresses to include investigations that control variables and provide evidence to support explanations or design solutions. • Conduct an investigation collaboratively to produce data to serve as the basis for evidence, using fair tests in which variables are controlled and the number of trials considered. (5-PS1-4) • Make observations and measurements to produce data to serve as the basis for evidence for an explanation of a phenomenon. (5-PS1-3) Using Mathematics and Computational Thinking Mathematical and computational thinking in 3–5 builds on K–2 experiences and progresses to extending quantitative measurements to a variety of physical properties and using computation and mathematics to analyze data and compare alternative design solutions. • Measure and graph quantities such as weight to address scientific and engineering questions and problems. (5-PS1-2) Crosscutting Concepts
Cause and Effect • Cause and effect relationships are routinely identified and used to explain change. (5-PS1-4)
Scale, Proportion, and Quantity • Natural objects exist from the very small to the immensely large. (5-PS1-1) • Standard units are used to measure and describe physical quantities such as weight, time, temperature, and volume. (5-PS1-2),(5-PS1-3)
Connections to Nature of Science
Scientific Knowledge Assumes an Order and Consistency in Natural Systems • Science assumes consistent patterns in natural systems. (5-PS1-2))
Performance Expectations
Students who demonstrate understanding can:
5-PS1-1. Develop a model to describe that matter is made of particles too small to be seen. [Clarification Statement: Examples of evidence supporting a model could include adding air to expand a basketball, compressing air in a syringe, dissolving sugar in water, and evaporating salt water.] [Assessment Boundary: Assessment does not include the atomic-scale mechanism of evaporation and condensation or defining the unseen particles.] 5-PS1-2. Measure and graph quantities to provide evidence that regardless of the type of change that occurs when heating, cooling, or mixing substances, the total weight of matter is conserved. [Clarification Statement: Examples of reactions or changes could include phase changes, dissolving, and mixing that form new substances.] [Assessment Boundary: Assessment does not include distinguishing mass and weight.] 5-PS1-3. Make observations and measurements to identify materials based on their properties. [Clarification Statement: Examples of materials to be identified could include baking soda and other powders, metals, minerals, and liquids. Examples of properties could include color, hardness, reflectivity, electrical conductivity, thermal conductivity, response to magnetic forces, and solubility; density is not intended as an identifiable property.] [Assessment Boundary: Assessment does not include density or distinguishing mass and weight.] 5-PS1-4. Conduct an investigation to determine whether the mixing of two or more substances results in new substances.
Common Core State Standards Connections
ELA/Literacy
RI.5.7 Draw on information from multiple print or digital sources, demonstrating the ability to locate an answer to a question quickly or to solve a problem efficiently. (5-PS1-1) W.5.7 Conduct short research projects that use several sources to build knowledge through investigation of different aspects of a topic. (5-PS1-2),(5-PS1-3),(5-PS1-4) W.5.8 Recall relevant information from experiences or gather relevant information from print and digital sources; summarize or paraphrase information in notes and finished work, and provide a list of sources. (5-PS1-2),(5-PS1-3),(5-PS1-4) W.5.9 Draw evidence from literary or informational texts to support analysis, reflection, and research. (5-PS1-2),(5-PS1-3),(5-PS1-4) Mathematics MP.2 Reason abstractly and quantitatively. (5-PS1-1),(5-PS1-2),(5-PS1-3) MP.4 Model with mathematics. (5-PS1-1),(5-PS1-2),(5-PS1-3) MP.5 Use appropriate tools strategically. (5-PS1-2),(5-PS1-3) 5.NBT.A.1 Explain patterns in the number of zeros of the product when multiplying a number by powers of 10, and explain patterns in the placement of the decimal point when a decimal is multiplied or divided by a power of 10. Use whole-number exponents to denote powers of 10. (5-PS1-1) 5.NF.B.7 Apply and extend previous understandings of division to divide unit fractions by whole numbers and whole numbers by unit fractions. (5-PS1-1) 5.MD.A.1 Convert among different-sized standard measurement units within a given measurement system (e.g., convert 5 cm to 0.05 m), and use these conversions in solving multi-step, real-world problems. (5-PS1-2) 5.MD.C.3 Recognize volume as an attribute of solid figures and understand concepts of volume measurement. (5-PS1-1) 5.MD.C.4 Measure volumes by counting unit cubes, using cubic cm, cubic in, cubic ft, and improvised units. (5-PS1-1)

Please Login or Subscribe to access downloadable content.
Citing Research References
When you research information you must cite the reference. Citing for websites is different from citing from books, magazines and periodicals. The style of citing shown here is from the MLA Style Citations (Modern Language Association).
When citing a WEBSITE the general format is as follows. Author Last Name, First Name(s). "Title: Subtitle of Part of Web Page, if appropriate." Title: Subtitle: Section of Page if appropriate. Sponsoring/Publishing Agency, If Given. Additional significant descriptive information. Date of Electronic Publication or other Date, such as Last Updated. Day Month Year of access < URL >.
Here is an example of citing this page:
Amsel, Sheri. "Grade 5 - 5-PS1 Matter and Its Interactions" Exploring Nature Educational Resource ©2005-2024. March 25, 2024 < http://www.exploringnature.org/db/view/Grade-5-5-PS1-Matter-and-Its-Interactions >
Exploringnature.org has more than 2,000 illustrated animals. Read about them, color them, label them, learn to draw them.


5 Experiments to Teach Chemical Changes to Elementary Students
Why you should teach chemical changes.
A chemical change occurs when a chemical substance is transformed into one or more other substances. I find that teaching chemical changes is a great way to teach the properties of matter because it provides students an opportunity to visually see a change, rather than just interacting with numbers and formulas. Often the best way to teach science is by bringing complex ideas to life and making it fun!
The following five experiments are my favorite ways to teach chemical changes to elementary students:
1. Observing the Formation of Gas
This is a very easy experiment to teach a chemical change to young kids. For this experiment, collect an old soda bottle, vinegar, baking soda, a balloon, and a funnel. Have the students put two spoonfuls of baking soda into the balloon and then using the funnel, pour vinegar into the old soda bottle until it is about half full. Stretch the balloon around the top of the bottle carefully to not let the baking soda fall into the bottle. When ready, make the balloon stand up by pouring the baking soda into the bottle.

When baking soda reacts with vinegar, a gas (carbon dioxide) is created. This gas is trapped inside the bottle and balloon, causing the balloon to inflate. Students can observe how this chemical change completely changes the balloon!
2. Observe Color Changes
First, fill three glass jars halfway with water. Separately, collect food coloring of 3 different colors, a cup of bleach, vinegar, and hydrogen peroxide (do not mix). Start the experiment by having students put colorings into the water to show a physical change (color change). Next, the teacher puts a spoonful of vinegar in one glass, a spoonful of bleach in another glass, and a spoonful of hydrogen peroxide in the last glass (and don’t forget safety glasses while you do!). Have the students determine in which glass(es) a physical change occurred and in which a chemical change occurred. This will open a discussion on how they knew which mixtures prompted physical or chemical changes, with the students providing real examples from their own observations.

3. Make Orange Fizz
Another super fun (and tasty) way to teach chemical changes is to make orange fizz. All you need are oranges and baking soda. Cut the orange and dip a slice into baking soda and try it!
4. Make Instant Snow
How much fun would it be to make snow in the summer to teach science? For this fun experiment, all you need is shaving cream and baking soda. Mix about 2 cups of each in a bowl and keep adding more to get the consistency that you like. Have fun and see the chemical change right in front of your eyes! Download the free activity sheet for full instructions.

5. Mentos and Coke
This is the most popular of all chemical change experiments of all! For this experiment, all you need is diet coke and mentos. Open the bottle of diet coke, quickly drop the mentos in, and back away quick. Kids will love watching the geyser that explodes! This always gets a great reaction out of the students and really brings chemical changes to life!
Looking for more chemistry ideas? Check out these engaging chemistry experiments!

About the Author: Jessica Fitzpatrick

Related Posts

Build an Island: Middle School Topography Lesson
Edible Rocks Experiment

STEM Valentine’s Day Activities

Intro to Ecology: 5 Free Worksheets
Winter Geometry Worksheets
Leave a comment cancel reply.
Save my name, email, and website in this browser for the next time I comment.
Sign up to receive the latest STEM resources, activities, and more from educational professionals like you straight to your inbox!
- Rating Count
- Price (Ascending)
- Price (Descending)
- Most Recent
Physical changes experiment
Resource type.

Types of Chemical Reactions Activity & Physical and Chemical Changes Experiments

Chemical and Physical Changes with Hands-On Experiments - 5.P.2.3

Physical and Chemical Changes Lab Activities and Experiments

Physical and Chemical Changes in Matter - Experiments , Activities and Posters

Physical and Chemical Changes Unit for Special Education with experiments

Physical & Chemical Changes Science: S'mores Lab Activity / Experiment

CER Slime Lab Chemistry Experiment - Physical & Chemical Changes

Diet Coke And Mentos Geyser Experiment | Physical Changes STEM | Outdoor Science

Physical vs Chemical Changes Science Unit Bundle - Experiments , Activities, etc.

Physical and Chemical Changes - Hands On Experiments

Candy Science | Physical Changes Gas Experiment | Matter And Interactions Lab

Physical Chemical Changes Labs Experiments Stations Packet NGSS

Making Ice Cream in a Bag Science Experiment | Chemical and Physical Change

Physical and Chemical Change - Experiments

Taste Science Chemistry | CER Lab Experiments | Physical & Chemical Change

Smores Physical /Chemical Change of Matter Experiment

Physical vs. Chemical Change & Properties - Lesson Plan, 2 Lab Experiments

Physical and Chemical Change Labs & Experiments for 4th & 5th Grade

Physical and Chemical Changes Experiment - Lab Rotation

Chemical VS Physical Change 5 Experiments [Sheets, How-to, Stations] Chemistry

Physical and Chemical Changes Scoot, Game, Experiments , Color Chemistry/Science

5th Grade Chemical & Physical Change Lesson / Experiments

Physical and Chemical Changes Activity | Elephants Toothpaste Experiment .

No Prep Online Physical & Chemical Changes Experiments Assignment

- We're hiring
- Help & FAQ
- Privacy policy
- Student privacy
- Terms of service
- Tell us what you think
Sciencing_Icons_Science SCIENCE
Sciencing_icons_biology biology, sciencing_icons_cells cells, sciencing_icons_molecular molecular, sciencing_icons_microorganisms microorganisms, sciencing_icons_genetics genetics, sciencing_icons_human body human body, sciencing_icons_ecology ecology, sciencing_icons_chemistry chemistry, sciencing_icons_atomic & molecular structure atomic & molecular structure, sciencing_icons_bonds bonds, sciencing_icons_reactions reactions, sciencing_icons_stoichiometry stoichiometry, sciencing_icons_solutions solutions, sciencing_icons_acids & bases acids & bases, sciencing_icons_thermodynamics thermodynamics, sciencing_icons_organic chemistry organic chemistry, sciencing_icons_physics physics, sciencing_icons_fundamentals-physics fundamentals, sciencing_icons_electronics electronics, sciencing_icons_waves waves, sciencing_icons_energy energy, sciencing_icons_fluid fluid, sciencing_icons_astronomy astronomy, sciencing_icons_geology geology, sciencing_icons_fundamentals-geology fundamentals, sciencing_icons_minerals & rocks minerals & rocks, sciencing_icons_earth scructure earth structure, sciencing_icons_fossils fossils, sciencing_icons_natural disasters natural disasters, sciencing_icons_nature nature, sciencing_icons_ecosystems ecosystems, sciencing_icons_environment environment, sciencing_icons_insects insects, sciencing_icons_plants & mushrooms plants & mushrooms, sciencing_icons_animals animals, sciencing_icons_math math, sciencing_icons_arithmetic arithmetic, sciencing_icons_addition & subtraction addition & subtraction, sciencing_icons_multiplication & division multiplication & division, sciencing_icons_decimals decimals, sciencing_icons_fractions fractions, sciencing_icons_conversions conversions, sciencing_icons_algebra algebra, sciencing_icons_working with units working with units, sciencing_icons_equations & expressions equations & expressions, sciencing_icons_ratios & proportions ratios & proportions, sciencing_icons_inequalities inequalities, sciencing_icons_exponents & logarithms exponents & logarithms, sciencing_icons_factorization factorization, sciencing_icons_functions functions, sciencing_icons_linear equations linear equations, sciencing_icons_graphs graphs, sciencing_icons_quadratics quadratics, sciencing_icons_polynomials polynomials, sciencing_icons_geometry geometry, sciencing_icons_fundamentals-geometry fundamentals, sciencing_icons_cartesian cartesian, sciencing_icons_circles circles, sciencing_icons_solids solids, sciencing_icons_trigonometry trigonometry, sciencing_icons_probability-statistics probability & statistics, sciencing_icons_mean-median-mode mean/median/mode, sciencing_icons_independent-dependent variables independent/dependent variables, sciencing_icons_deviation deviation, sciencing_icons_correlation correlation, sciencing_icons_sampling sampling, sciencing_icons_distributions distributions, sciencing_icons_probability probability, sciencing_icons_calculus calculus, sciencing_icons_differentiation-integration differentiation/integration, sciencing_icons_application application, sciencing_icons_projects projects, sciencing_icons_news news.
- Share Tweet Email Print
- Home ⋅
- Science Fair Project Ideas for Kids, Middle & High School Students ⋅
5th Grade Chemical Change Activity

Simple Chemical Change Experiments for 4th Graders
A chemistry project for 5th graders should appear more like fun and less like learning. Illustrating a chemical reaction by changing the color of a penny fits the bill. This is an experiment that a 10-year old can do virtually on his own, and is one that delivers immediate as well as long term results. The variety of "chemicals" used for this project will yield an array of colors.
Explaining the Chemical Reaction

Pennies are made with a zinc core that has been coated in a thin layer of copper. Different types of chemicals react with the copper to cause discoloration; mostly in some shade of green. A good example is the Statue of Liberty. It is coated in copper but has turned green over the years as a reaction to the elements. Phosphoric, carbonic, citric and acetic acids will all react differently to the copper on the penny.
Setting Up the Experiment
You will need four clear plastic cups, four discolored pennies, cola, lemon juice, salt and vinegar. Place a penny in the bottom of each of the four cups. Pour cola over one penny just enough to cover it. Repeat this with the lemon juice and twice with the vinegar. Sprinkle about a quarter teaspoon of salt into one of the vinegar cups. Have the child label the cups "phosphoric/carbonic acid" for the cola, "citric acid" for the lemon juice, "acetic acid" for the the vinegar and "acetic acid with salt" for the salt and vinegar cup.
Recording the Results
Once you begin to see a penny begin to change, have your child make a note of it. Keeping a record of results is a critical step in the chemical change. The most effective way to log results for this experiment is first by cup, then by time. Have your child make four columns: one for each cup, then list the time and result as the pennies begin to change.
Ask Questions
Engage your child by talking about the results. Ask questions such as, "Which one began to change first?" and "Which change were you most surprised by? Why?" Considering this chemical change activity can take more than 24 hours to complete, you will have plenty of time for observation and interaction.
Related Articles
Chemical reactions to do at home, how to calculate an average percent change, experiments with salt and vinegar, food coloring & science projects, five characteristics of a chemical change, how to do cool science experiments with rubbing alcohol..., simple photosynthesis activities, coin corrosion science experiments for kids, why do pennies change color, chemical vs. physical reactions, color-changing liquid experiments, penny science fair project ideas, seven things that indicate a chemical change is occurring, what is the overall order of reaction, how to find the molar heat of a combustion candle, 5 ways to know if a chemical change has occurred, experiments on cleaning pennies, characteristics of chemical reactions, at-home science: color mixing experiment.
- A Green Penny??: Knight Foundation Summer Institute
- I Love Bacteria: Colored Pennies
About the Author
Since 1995, Sharideth Smith has written everything from 400-word blog posts to political platforms. Her work has been featured on various online publications and she has a solid following on her own website where she has been doling out relationship advice since 2009.
Photo Credits
Photodisc/Photodisc/Getty Images
Find Your Next Great Science Fair Project! GO

IMAGES
VIDEO
COMMENTS
These ideas aren't your average science experiments! We're going to dig deeper with conservation of matter, CER, and a phenomenon-based science unit. There are MANY physical and chemical changes you can observe, but here are 5 of my favorites for upper elementary. Many of these ideas include a focus on conservation of matter because the ... title="5 Ideas for Physical & Chemical Change ...
Chemical and Physical Changes Experiment #3: Gas Balloons. Another fun but easy experiment to use in your classroom involves soda and a balloon. Take a 1-liter bottle of any brand of soda and place a balloon around the mouth of the bottle. This experiment will need about ten minutes to see the changes. Over time, students should observe that ...
Chemical changes make new substances. Anytime a new substance is made, a chemical change takes place. Usually two or more materials are combined and a new substance is formed. A chemical change can produce amazing explosions, like fireworks. Some chemical changes are a little more difficult to spot, like when a nail rusts.
PE 5 Integrated Curriculum Grade 5: Science (Draft 2023) DK 1.5: ... To help students keep track of the experiment over the course of the week, use the Physical and Chemical Change Experiments Predict and Observe Chart reproducible to monitor the changes day by day. Language. Consider creating a word wall of terminology that students will ...
Physical Change. The key thing to understand when learning about physical changes is that physical changes do not make a new substance. The definition of physical change is a change in the shape or appearance of an object, like crumpling a piece of paper, or cutting, bending, or dissolving something. Physical changes also happen when matter ...
Are you looking for fun and easy physical and chemical changes activities for 5th grade? You can do these experiments at home or in the classroom. I will also show you some great sketch notes and worksheets covering matter, properties of matter, and physical and chemical changes. Affiliate links for Amazon are included. Activities for Physical ...
Physical changes you may notice; changes in volume, texture (wet or dry), size, color. What Melts. Compare how fast different everyday items melt in the sun, including ice cubes. A fun experiment to do in the summer! Physical Change Experiments That Look Like Chemical Reactions. The science experiments below are all examples of physical change.
Step 2. Chemical Change: In a chemical change a new substance is formed and energy is either given off or absorbed. To explore chemical change, we performed a number of experiments. First, w ith an adult present, crumple the paper towel back into a ball and use a lighter or match to light it on fire. Quickly drop it into a glass.
Add the salt to the water and mix the solution with a spoon. Be sure to not lose any of the salt or water during the mixing. Find the mass of the cup, salt-water mixture, and the empty salt container. ** The mass should not change because mass is conserved. Experiment IV. In a chemical change, mass is again conserved.
Grade 5: Matter that changes state is still the same matter. Physical change refers to the fact that a substance can be changed from one form to another. Chemical change implies the formation of a new substance. Materials if dropping off at a school (per student unless otherwise noted).
Physical and Chemical Change Experiments Graduate Fellow: Megan Fencil Master Teacher: Ada Anderson Grade level: 5th Objective: Students will conduct a series of experiments to learn the difference between physical and chemical changes. TEKS: 5.1a, 5.2a-d, 5.4a, 5.7a-d Time Allotment: 80 - 90 minutes Materials: Safety goggles Graduated cylinders
It is essential to know the difference between the two ways that matter can change. Physical Change: Changes the appearance of matter but does NOT create a new substance. Ex. Changes in the size, shape, or state of matter. Chemical Change: A change that creates a new substance. Teach students that there are four ways to identify when matter ...
The Grade 5 Physical Science Unit is presented to students through a series of investigations, experiments, active learning experiences, questions, and assessments. Assessments include: pre-, post-, and 4 formative assessments. ... In Lesson 5, "Physical Changes, ...
Observing the Formation of a Gas. How a Chemical Reaction Can Produce Heat. The Formation of a Solid. The Occurrence of a Color Change. The Elephant Toothpaste Experiment. Hot Ice. Mentos and Coke Experiment. Separate a Mixture. One note before I move on to the activities.
You can incorporate chemical and physical reaction learning into snack time! Learners often think physical changes are reversible, however, popcorn is the perfect example of a physical change that we cannot return to the original kernel state. Discuss as you make this snack! Learn More: Edventures with Kids. 22. Get the Scoop on Dairy Products
Matter is capable of undergoing changes, which are classified as either physical or chemical. Physical changes in matter are often reversible. For example, an ice cube can melt into liquid water, and then the liquid water can be refrozen into an ice cube. A chemical change is very different. A burning log demonstrates this.
These chemical change labs explore five types of evidence: bubbles, color, heat, odor, and light. First, kids experiment with physical change. Then they launch into reactions that create new substances. Follow my TPT store for updates.
Fifth Grade Physical Science Activities. Study Game: Planets! All you need are pencils, paper and some scissors to get started on this easy and fun game to review astronomy information, or other types of study concepts. Click the checkbox for the options to print and add to Assignments and Collections.
RI.5.7 Draw on information from multiple print or digital sources, demonstrating the ability to locate an answer to a question quickly or to solve a problem efficiently. (5-PS1-1) W.5.7 Conduct short research projects that use several sources to build knowledge through investigation of different aspects of a topic. (5-PS1-2),(5-PS1-3),(5-PS1-4)
The science stations cover all the appropriate concepts for Chemical Reactions. Everything your fifth-graders need to know about Chemical Reactions is included. These eight different science stations all teach and incorporate important skills through labs, experiments, videos, text, and games. Best of all, your students will not only understand ...
The following five experiments are my favorite ways to teach chemical changes to elementary students: 1. Observing the Formation of Gas. This is a very easy experiment to teach a chemical change to young kids. For this experiment, collect an old soda bottle, vinegar, baking soda, a balloon, and a funnel. Have the students put two spoonfuls of ...
Teaching Muse. 5.0. (59) $4.00. Zip. Reinforce characteristics and concepts related to physical and chemical changes with these 8 hands-on lab activity experiments. Each activity will further help students learn about physical and chemical changes. Students will make observations and demonstrate their knowledge by making predictions and ...
Updated April 24, 2017. By Sharideth Smith. A chemistry project for 5th graders should appear more like fun and less like learning. Illustrating a chemical reaction by changing the color of a penny fits the bill. This is an experiment that a 10-year old can do virtually on his own, and is one that delivers immediate as well as long term results.